Genetics is becoming a factor in the diagnosis and treatment of all kinds of disorders. While aware of medical genetics' growing importance, many people, both doctors and patients, feel it is difficult to keep pace with this rapidly advancing area. Famed geneticist, Dr. Jim Lupski, outlines the basic terrain of the genome project and introduces some of its issues and vocabulary.
— The Editors
— The Editors
Birth defects are the leading cause of death among infants (0-364 days old). By age 25, eight percent of liveborns will be diagnosed with a disorder that has a major genetic component. The goals of the human genome project are to identify all genes in the human genome, determine their structure and function and determine their involvement in human disease. Let's see if we can walk through some of the basic concepts and terminology.
Mutation and Human Genetic Disease
As mentioned, 8% of liveborns will suffer from a genetically based disorder by age 25. We refer to the all these genetic defects as the genetic load. Genetic load is produced by five paths:
- Single gene disorders account for 3.6/1000 (.36%)
autosomal dominant 1.4/1000 (.14%)
autosomal recessive 1.7/1000 (.17%)
X-linked recessive 0.5/1000 (.05%) - Chromosome abnormalities account for 1.8/1000 (.18%)
- Multifactorial disorders are believed to account for 46.4/1000 (4.64%)
- Genetic unknown=0.12%
- Congenital anomalies with a genetic etiology = 2.66%
(all congenital anomalies = 5.25%)
Genome Mutations
The genome is defined as the total genetic material contained within the chromosomes of an organism. Humans have 46 chromosomes, 23 from each parent. Our 46 chromosomes represent our genome.
Genome mutations occur at a frequency of about 1/10,000 to 1/100,000. Polyploidy occurs when an extra copy of every chromosome is made, trisomy when only an extra copy of a single chromosome occurs. Trisomy 21 (Down syndrome) is perhaps the most frequently observed genetic disease, occurring in 1:660 newborns. If an entire chromosome is absent, most frequently in Turner syndrome, the disorder is called a monosomy.
Chromosome Mutations
Structural rearrangements in chromosomes that can be detected microscopically by changes in the banding patterns produced by dye specific staining of chromosomes are said to be chromosomal mutations. There are several major types:
Microdeletions
involve deletion of a small portion of a chromosome, sometimes several contiguous genes, produce a set of syndromes which are now being appreciated to a much greater extent.
Microduplications
produce an extra exact copy of a specific region of a specific chromosome.
Inversions
re-order a segment of chromosome in backwards order.
Translocations
occur when a piece of a chromosome attaches to another chromosome. Individuals who harbor these translocations are said to be balanced (because they have the proper amount of genetic information) but their offspring can, as the result of incorrect attachment producing duplications and deletions, be unbalanced. It is estimated that one in every 550 individuals on this earth may contain a balanced translocation. If three or more miscarriages occur in a family, unbalance should be suspected and the family's chromosomes investigated.
Gene Mutations
Genes are the most basic unit of heredity and are built from DNA. The DNA molecule is constructed from two vertical strands of chemicals (bases) — adenine (A), guanine (G), cytosine (C) and thymine (T) — coiled around each other like a twisted ladder (the double helix). Each parent contributes one of the vertical strands. The DNA ladder is held together by rungs formed by bonds between bases (base pairs). Thousands of base pairs are needed for each gene. It is believed that the human genome has between 50,000 and 100,000 genes. Gene mutations occur in a frequency believed to be between 1/100,000 to 1/1,000,000.
DNA rearrangments
Deletions in portions of a gene may occur and usually less than 20 base pairs may be involved.
Duplications are exact copies of a DNA sequence.
Substitutions involve replacement of one sequence fragment with another sequence.
Insertions can occur by the movement of specific sequences and insertion into a gene. Examples include AluI repetitive sequence insertion leading to neurofibromatosis type I and hemophilia.
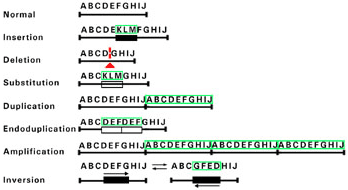
Figure 1. DNA rearrangements. Mutations resulting from DNA rearrangements usually involve gross loss or rearrangement of many base pairs. The letters are used to depict several DNA bases. Below each set of letters are lined drawings of the DNA sequences. Each alteration in DNA sequence resulting from a particular rearrangement is surrounded by green lines. The triangle denotes a deletion.
Point Mutations
A point mutation is a change affecting a single position in a gene. They are thought to arise by chemical, physical or enzymatic mechanisms. The efficiency of these mechanisms is known to be sequence dependent, that is, they are nonrandom and occur at so-called mutation hot spots.
Transitions are changes of a purine base [adenine (A) or guanine (G)] to another purine (A→G or G→A) or of a pyrimidine base [cytosine (C) or thymine (T)] to another pyrimidine (C→>T or T→C). They occur much more frequently than transversions (purine change to pyrimidine or pyrimidine change to purine). Transitions occur 14 times more frequently than transversions.
Mutations can be silent and not change the coding of a specific amino acid, while missense mutations lead to a different amino acid substitution. Nonsense mutations are point mutations that change a triplet codon (three base pairs) encoding a specific amino acid. Frameshift mutations delete bases of greater than or less than three in number. This shifts the reading frame and produces a subsequent stop codon.…[T]he chromosome involved in Down syndrome, for example, represents 1.9% of the three billion base pairs that make up the human genome.
Most of the point mutations, which have been found to cause human genetic disease, occur as the result of misincorporation of bases during DNA replication.
Detecting a Mutation
Remember we have 46 chromosomes and each chromosome is made of many genes and each gene is constructed from base pairs. As we discussed, a mutation can be caused by a change in a whole chromosome or involve just one base pair of a specific gene. Therefore, doctors have developed many different methods appropriate to the physical size of the mutation. Chromosome 21, the chromosome involved in Down syndrome, for example, represents 1.9% of the three billion base pairs that make up the human genome. If it were possible to examine the 57 million base pairs on Chromosome 21, which is not technically practical or possible, the doctor would still not be able to find the mutation responsible for Down syndrome because the problem isn't at the gene level. The Down patient has three chromosome 21's instead of the normal two copies. There are, in other words, no mutant genes in individuals with Down syndrome, just an extra copy of the whole chromosome. The extra chromosome occurs because of an error at the time the embryonic egg is made. This "segregation" error more frequently occurs in aging females, thus the increased chance of these disorders with advanced maternal age.
In contrast, a patient with sickle cell disease has a change in only one base pair out of the three billion in their genome. You could look forever at the 46 chromosomes and would never see any abnormal chromosomes. To find the genetic error, the doctor would have to sequence the DNA, that is, determine the order of the base pairs on the globin gene of the patient with sickle cell disease and note how it differs from the normal. For sickle cell disease, scientists have done this and identified the one base pair change, but for many other diseases the exact genetic change has not yet been detected.
The Benefits of the Human Genome Project
It is estimated that the sequence of the human genome should be completely mapped by approximately the year 2005. Already, however, the many new techniques developed so far during this project have provided doctors with improved genetic diagnostics and predictive testing.
The availability of extensive genetic maps has increased the pace by which different disease genes are localized in the human genome. The techniques have also enabled doctors to identify susceptible areas of the genome that may be responsible for some disorders, such as diabetes, hypertension and certain forms of cancer that are more complicated and are caused by more than one genetic change.




